L'Ittero Neonatale
L’ittero neonatale è una condizione frequente, causata da alti livelli di bilirubina nel sangue. Se non curato in tempi brevi può determinare l’insorgere del kernittero, patologia che provoca danni neurologici permanenti.

L'Individuazione Dei Casi
Per quantificare il livello di bilirubina in un neonato attualmente è necessario fare un prelievo di sangue, intervento invasivo eseguito solo da personale specializzato, presso strutture sanitarie ospedaliere.

Le Necessità
Sviluppare un sistema che preveda l’uso di una tecnica non invasiva, che possa garantire test semplici, rapidi e affidabili, utilizzabile anche al di fuori dalle strutture ospedaliere, è diventato sempre più necessario.

La Nostra Soluzione
Il Sistema Bilistick® è in grado di produrre risultati per la diagnosi immediata, con attendibilità e affidabilità dei test paragonabili a quelle dei metodi di laboratorio, a partire da una goccia di sangue del neonato.
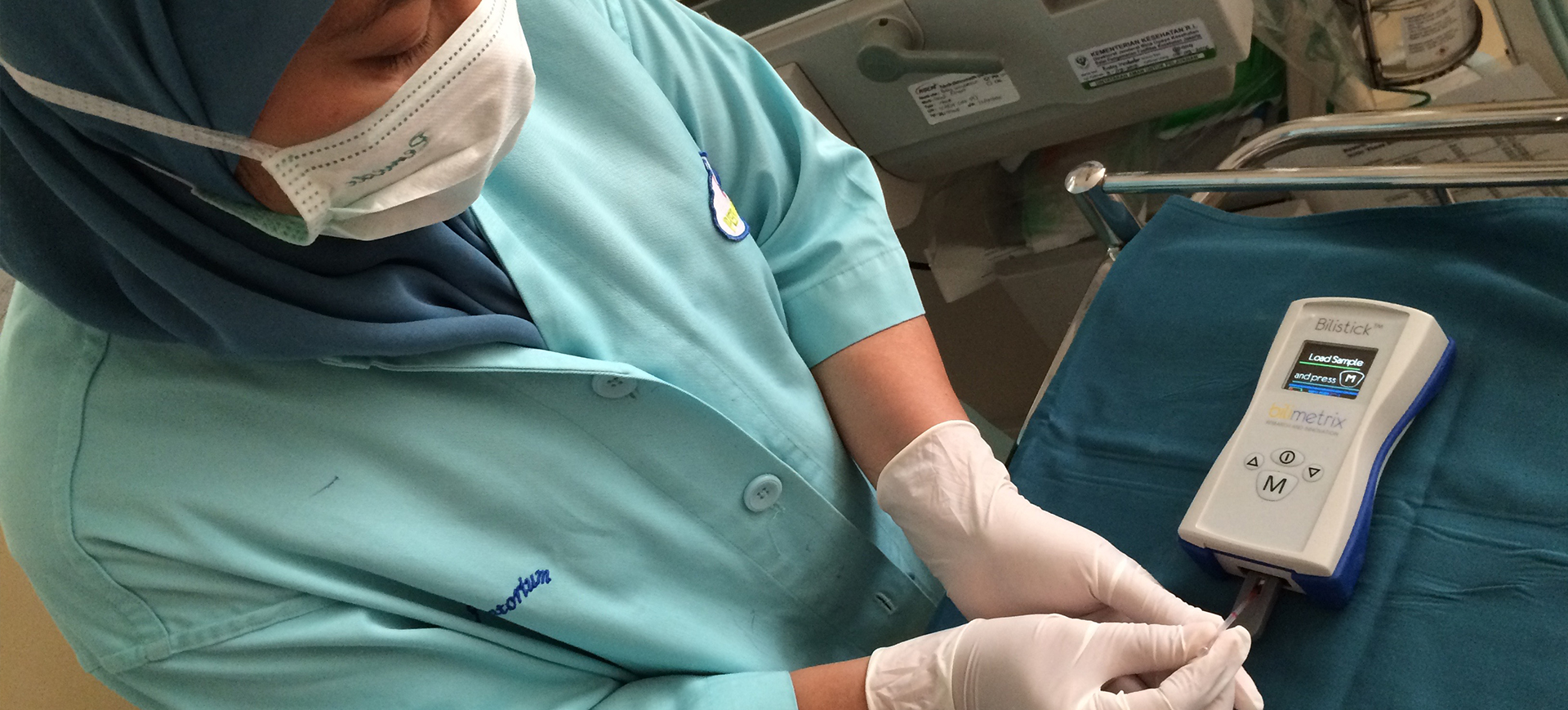
Il Sistema Bilistick®
Come Funziona
Il Sistema Bilistick® ideato e realizzato da Bilimetrix consente la misurazione della bilirubina nel sangue attraverso un processo poco invasivo, allo scopo di determinare l'eventuale presenza di ittero neonatale. Il sistema è molto semplice da utilizzare: basta inserire la Striscia di Test Bilistick® nel Lettore Bilistick® e depositare su di essa una piccola goccia di sangue. In questo modo è possibile ottenere la misurazione della bilirubina sierica in meno di due minuti con un’accuratezza comparabile a quella dei metodi tradizionali di laboratorio.
Il Team Bilimetrix

Diego Sardon
Presidente e Amministratore Delegato

Carlos Coda Zabetta
Direttore Tecnico e Responsabile Aspetti Regolatori

Chiara Greco
Specialista Aspetti Tecnici e Responsabile Qualità

Cristina Fernetti
Responsabile Produzione
Principali Riconoscimenti
RICONOSCIMENTI A LIVELLO NAZIONALE
RICONOSCIMENTI A LIVELLO EUROPEO E INTERNAZIONALE
Pubblicazioni E Presentazioni
Ultime notizie